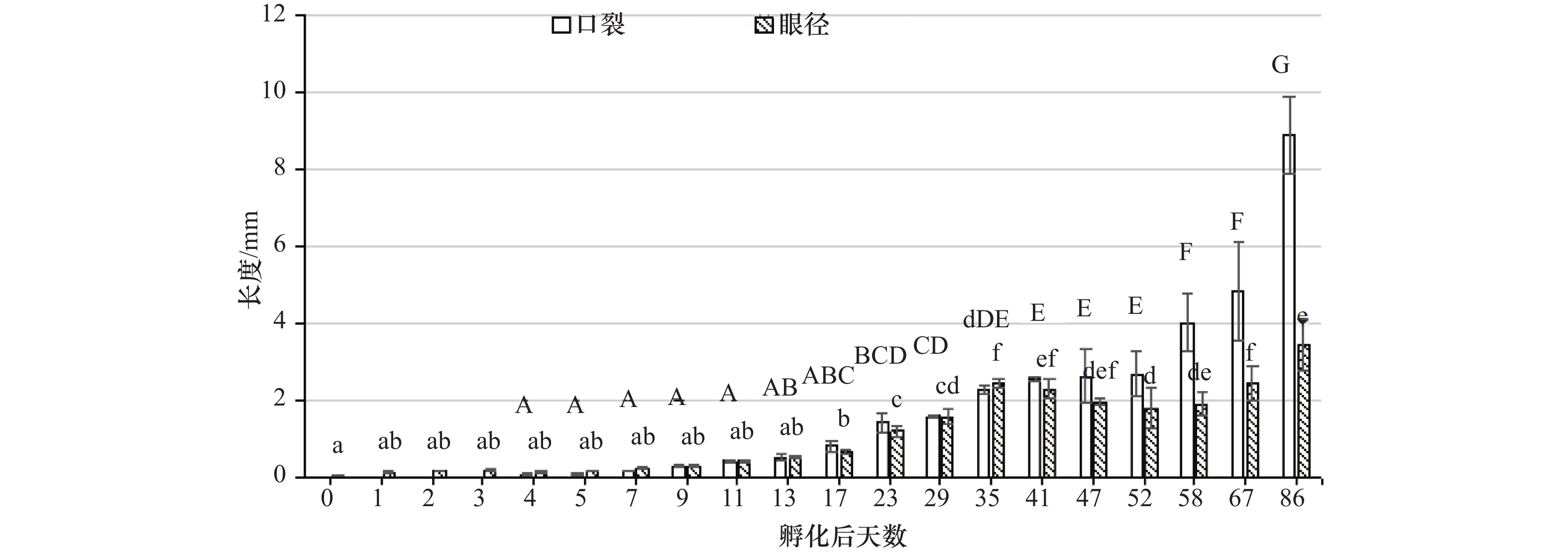
Analysis of metamorphosis development and growth characteristics of hybrid offspring of Epinephelus lanceolatus ♀×Epinephelus. tukula ♂
-
摘要: 为了深入研究鞍带石斑鱼(Epinephelus lanceolatus) (♀)×蓝身大斑石斑鱼(E. tukula) (♂)杂交F1代的变态发育及生长特征,本研究对杂交组和纯种鞍带石斑鱼胚胎发育时间、受精率、畸形率、孵化率和仔稚幼鱼生长性状(全长、体长、体高、肛前距),以及杂交组卵黄囊和油球吸收过程,第二背鳍棘和腹鳍棘生长和收缩,口径和眼裂的变化等进行了详细的观察描述和统计分析。结果显示,在水温28℃条件下,杂交组和鞍带石斑鱼受精卵分别经21 h 24 min和21 h 32 min完成胚胎发育;杂交F1代受精率、畸形率和孵化率分别是89.09%±0.08%、35.16%±5.05%和62.59%±10.70%,与纯种鞍带石斑鱼无显著性差异。根据卵黄囊、第二背鳍棘、腹鳍棘、鳞片、体色等形态变化,将其胚后变态发育分为前期仔鱼(孵化后0~6 d)、后期仔鱼(孵化后7~34 d)、稚鱼期(孵化后35~46 d)和幼鱼期(孵化后47~86 d),前期仔鱼生长较缓慢,后期仔鱼到幼鱼期生长逐渐加快,86 d时杂交组和对照组体长分别达(60.80±0.50) mm和(51.80±0.47) mm,杂交组生长速度极显著高于对照组(P<0.01)。在胚胎发育时期卵黄囊消耗量为29.45%,油球消耗量为20.75%,卵黄囊在孵化后1 d消耗最快,达58.70%。油球在孵化后3 d消耗最快,达32.08%,孵化后第5 d仔鱼卵黄囊和油球基本吸收完毕。第二背鳍棘、腹鳍棘在孵化后29 d达到最长,分别是(8.15±0.02) mm和(5.80±0.10) mm,至47 d完全退化,变态发育完成。第一天仔鱼眼径为(0.16±0.01) mm,至86 d眼径增大22倍,第四天口裂长为(0.09±0.02) mm,至86 d增大99倍。结果表明,鞍带石斑鱼(♀)×蓝身大斑石斑鱼(♂)杂交F1代胚胎和仔稚幼鱼发育正常,而且杂交F1代与母本相比具有较显著的生长优势,本研究为两种石斑鱼杂交苗种培育、发育研究和品种改良提供了丰富的数据。Abstract: In order to further study the metamorphosis development and growth characteristics of the hybrid offspring of Epinephelus lanceolatus♀×E. tukula♂, we measured embryonic development time, fertilization rate, deformity rate, hatching rate and growth traits (full length, body length, height and anal front) of the hybrid offspring and E. lanceolatus. We also observed the absorption process of yolk sac and oil globule, the growth and contraction of the second dorsal spine and pelvic fin spine, and the changes of eye diameter and oral fissure during embryonic development of hybrid offspring. Statistical analysis of the data was performed. The results show that the hybrid offspring and E. lanceolatus complete embryo development at 21 h 24 min and 21 h 32 min at 28℃, respectively; the fertilization rate, deformity rate and hatching rate of hybrid offspring are 89.09%±0.08%, 35.16%±5.05% and 62.59%±10.70%, respectively, no significant difference is shown compared with that of E. lanceolatus. According to the morphological changes of yolk sac, second dorsal spine, pelvic fin spine, scales and body color, the post-embryonic metamorphosis is divided into early larvae (0–6 d after hatching), late larvae (7–34 d after hatching), juveniles (35–46 d after hatching) and juvenile fish period (47–86 d after hatching). The growth of the early larvae is slower, while the growth of the larvae to the juveniles increases gradually. At 86 d after hatching, the body lengths of the hybrid offspring and E. lanceolatus reach (60.80±0.50) cm and (51.80±0.47) cm, which indicates the growth of the hybrid offspring is significantly faster than E. lanceolatus (P<0.01). During embryo development, the yolk sac consumption is 29.45%, the oil ball consumption is 20.75%, and the yolk sac consumption is the fastest at 1 d after hatching, reaching 58.70%. The oil globule consumption is the fastest at 3 d after hatching, reaching 32.08%, and the yolk sac and oil globule of the larvae are basically absorbed at 5 d after hatching. The lengths of the second dorsal spine and pelvic fin spine reach the longest of (8.15±0.02) mm and (5.80±0.10) mm at 29 d after hatching respectively, and they completely degenerate at 47 d after hatching which marks the completion of metamorphosis. The eye diameter of the larvae is (0.16±0.01) mm at 1 d after hatching, which increases by 22 times at 86 d after hatching. The length of oral fissure is (0.09±0.02) mm, and it increases by 99 times at 86 d after hatching. The results show that the hybrid offspring of E. lanceolatus♀×E. tukula♂ develops normally, and the hybrid offspring shows a significant growth advantage comparing with its female parent. This study provides rich data for seed cultivation, development research and variety improvement of the hybrid offspring.
-
图 1 鞍带石斑鱼(♀)×蓝身大斑石斑鱼(♂)杂交后代胚后发育
1.初孵仔鱼; 2. 1 日龄仔鱼;3. 2 日龄仔鱼;4.3日龄仔鱼;5. 4 日龄仔鱼;6.5日龄仔鱼;7. 7 日龄仔鱼;8. 9日龄仔鱼;9. 11 日龄仔鱼;10. 13 日龄仔鱼;11. 17日龄仔鱼;12. 23 日龄仔鱼;13.29日龄仔鱼;14.35 日龄稚鱼;15.41日龄稚鱼;16. 47 日龄幼鱼;17.52 日龄幼鱼;18.58日龄幼鱼; 19. 67日龄幼鱼;20. 86日龄幼鱼
Fig. 1 The development characteristics of hybrid Epinephelus lanceolatus (♀) × E. tukula (♂)
1. Newly hatched; 2. 1 d larvae; 3. 2 d larvae; 4.3 d larvae; 5. 4 d larvae; 6. 5 d larvae; 7. 7 d larvae; 8. 9 d larvae; 9. 11 d larvae; 10. 13 d larvae; 11. 17 d larvae;12.23 d larvae;13.29 d larvae; 14.35 d juvenile; 15. 41 d juvenile; 16. 47 d young fish; 17. 52 d young fish;18.58 d young fish;19.67 d young fish;20.86 d young fish
表 1 鞍带石斑鱼(♀)×蓝身大斑石斑鱼(♂)杂交F1代及对照组受精率、孵化率和畸形率
Tab. 1 Fertilization rate,hatching rate and deformity rate of E. lanceolatus (♀) × E. tukula (♂) and the control
组别 受精率 畸形率 孵化率 鞍带石斑鱼(♀)×蓝身大斑石斑鱼(♂) 89.09%±0.08%a 35.16%±5.05%b 62.59%±10.70%c 鞍带石斑鱼(♀)×鞍带石斑鱼(♂) 92.66%±1.68%a 27.79%±2.50%b 71.30%±2.50%c 注:相同字母表示性状之间无显著性差异(n=3, P<0.05)。 表 2 鞍带石斑鱼(♀)×蓝身大斑石斑鱼(♂)卵黄囊和油球的体积变化
Tab. 2 The volume change of yolk sac and oil absorption of E. lanceolatuss (♀) × E.tukula (♂)
日龄 全长/mm 卵黄囊长径/mm 卵黄囊短径/mm 油球直径/mm 卵黄囊体积/mm3 油球体积/mm3 受精卵 — 0.79±0.01 0.79±0.01 0.22±0.004a4 0.255 7±0.016 6a5 0.005 3±0.000 3a6 0 1.59±0.02a1 1.14±0.09a2 0.55±0.01a3 0.2±0.01b4 0.180 4±0.014 6b5 0.004 2±0.000 9b6 1 2.46±0.01b1 0.74±0.04b2 0.28±0.04b3 0.18±0.01c4 0.030 3±0.008 5c5 0.003 1±0.000 4c6 2 2.59±0.01c1 0.24±0.01c2 0.18±0.01c3 0.17±0.01c4 0.008 6±0.000 1d5 0.002 6±0.000 2c6 3 2.62±0.01c1 0.16±0.01c2 0.15±0.01d3 0.12±0.01d4 0.001 9±0.000 01d5 0.000 9±0.000 1d6 4 2.63±0.05cd1 0.15±0.01c2 0.12±0.01d3 0.11±0.01d4 0.001 2±0.000 2d5 0.000 7±0.000 2de6 5 2.68±0.08d1 0.13±0.05c2 0.10±0.01d3 0.05±0.01e4 0.000 7±0.000 6d5 0.000 1±0.000 006e6 注:数值为平均数±标准差(n=6);不同字母有显著性差异(n=10, P<0.05)。 -
[1] Xu Kang, DuanWei, Xiao Jun, et al. Development and application of biological technologies in fish genetic breeding[J]. Science China Life Sciences, 2015, 58(2): 187−201. doi: 10.1007/s11427-015-4798-3 [2] Arai K. Developmental genetic studies on salmonids: morphogenesis, isozyme phenotypes and chromosomes in hybrid embryos[J]. Memoirs of the Faculty of Fisheries-Hokkaido University, 1984, 30: 1−93. [3] Babiak I, Dobosz S, Kuzminski H, et al. Failure of interspecies androgenesis in salmonids[J]. Journal of Fish Biology, 2002, 61(2): 432−447. doi: 10.1111/jfb.2002.61.issue-2 [4] 吴清江, 付洪拓, 叶玉珍. 酶的基因剂量效应及其对鱼类远缘杂交的影响[J]. 水生生物学报, 1997, 21(2): 143−151. doi: 10.3321/j.issn:1000-3207.1997.02.007Wu Qingjiang, Fu Hongtuo, Ye Yuzhen. Effect of enzymic gene dosage on the distant hybridization of fishes[J]. Acta Hydrobiologica Sinica, 1997, 21(2): 143−151. doi: 10.3321/j.issn:1000-3207.1997.02.007 [5] 孙小异. 建鲤——我国养殖鱼类杂交选育的第一个品种[J]. 科学养鱼, 1988(4): 16−17.Sun Xiaoyi. Jian carp—the first breed of hybrid breeding of farmed fish in China[J]. Scientific Fish Farming, 1988(4): 16−17. [6] 楼允东. 鱼类育种学[M]. 北京: 中国农业出版社, 2001: 40–140.Lou Yundong. Fish Breeding[M]. Beijing: China Agriculture Press, 2001: 40– 140. [7] 吴水清, 郑乐云, 黄种持, 等. 云纹石斑鱼(♀)×鞍带石斑鱼(♂)杂交子代胚胎发育及仔稚幼鱼形态观察[J]. 渔业研究, 2016, 38(1): 27−35.Wu Shuiqing, Zheng Leyun, Huang Zhongchi, et al. Embryonic and morphological development in larva, juvenile, and young stages of hybrid grouper (Epinephelus moara ♀ ×E. lanceolatus ♂)[J]. Journal of Fisheries Research, 2016, 38(1): 27−35. [8] 陈超, 孔祥迪, 李炎璐, 等. 棕点石斑鱼(♀)×鞍带石斑鱼(♂)杂交子代胚胎及仔稚幼鱼发育的跟踪观察[J]. 渔业科学进展, 2014, 35(5): 135−144. doi: 10.11758/yykxjz.20140520Chen Chao, Kong Xiangdi, Li Yanlu, et al. Embryonic and morphological development in the larva, juvenile, and young stages of Epinephelus fuscoguttatus (♀) ×E. lanceolatus (♂)[J]. Progress in Fishery Sciences, 2014, 35(5): 135−144. doi: 10.11758/yykxjz.20140520 [9] 田永胜, 唐江, 马文辉, 等. 蓝身大斑石斑鱼冷冻精子(♂)与棕点石斑鱼卵(♀)杂交后代发育及生长特征分析[J]. 渔业科学进展, 2019, doi: 10.19663/j.issn2095-9869.20181017002.Tian Yongsheng, Tang Jiang, Ma Wenhuiu, et al. Development and growth of hybrid offspring brown grouper Epinephelus fuscoguttatus (♀)× Blue Speckled Grouper Epinephelus tulcula (♂) by using cryopreserved sperm[J]. Progress in Fishery Sciences, 2019, doi: 10.19663/j.issn2095-9869.20181017002. [10] 杨求华, 黄种持, 郑乐云, 等. 云纹石斑鱼(♀)×赤点石斑鱼(♂)杂交子代胚胎发育及生[J]. 海洋渔业, 2014, 36(3): 224−231. doi: 10.3969/j.issn.1004-2490.2014.03.005Yang Qiuhua, Huang Zhongchi, Zheng Leyun, et al. Embryonic development and growth of hybrid from the hybridization of Eqinephelus moara (♀) ×E. akaara (♂)[J]. Marine Fisheries, 2014, 36(3): 224−231. doi: 10.3969/j.issn.1004-2490.2014.03.005 [11] 刘付永忠, 赵会宏, 刘晓春, 等. 赤点石斑鱼♂与斜带石斑鱼♀杂交的初步研究[J]. 中山大学学报: 自然科学版, 2007, 46(3): 72−75.Liu Fuyongzhong, Zhao Huihong, Liu Xiaochu, et al. Preliminary study on the hybrid red-spotted grouper (Epinephelus akaara)♂×orange-spotted grouper (Epinephelus coioides)♀[J]. Acta Scientiarum Naturalium Universitatis Sunyatseni, 2007, 46(3): 72−75. [12] Glamuzina B, Kožul, Tutman P, et al. Hybridization of Mediterranean groupers: Epinephelus marginatus♀×E. aeneus♂ and early development[J]. Aquaculture Research, 1999, 30(8): 625−628. doi: 10.1046/j.1365-2109.1999.00365.x [13] James C M, Al-Thobaiti S A, Rasem B M, et al. Potential of grouper hybrid (Epinephelus fuscoguttatus ×E. polyphekadion) for aquaculture[J]. Naga, the ICLARM Quarterly, 1999, 22(1): 19−23. [14] 田永胜, 陈张帆, 段会敏, 等. 鞍带石斑鱼冷冻精子与云纹石斑鱼杂交家系建立及遗传效应[J]. 水产学报, 2017, 41(12): 1817−1828.Tian Yongsheng, Chen Zhangfan, Duan Huimin, et al. The family line establishment of the hybrid Epinephelus moara (♀)×E. lanceolatus (♂) by using cryopreserved sperm and the related genetic effect analysis[J]. Journal of Fisheries of China, 2017, 41(12): 1817−1828. [15] Apstein C. Die Bestimmung des alters pelagisch lebender fischeier[J]. Mitteilungender Deutschen Mathematiker-Vereinigung, 1909, 25: 364−373. [16] Tian Yongsheng, Jiang Jing, Wang Na, et al. Sperm of the giant grouper: cryopreservation, physiological and morphological analysis and application in hybridizations with red-spotted grouper[J]. Journal of Reproduction and Development, 2015, 61(4): 333−339. doi: 10.1262/jrd.2014-087 [17] 舒琥, 魏秋兰, 罗丽娟, 等. 广东沿海4种石斑鱼的染色体组型分析[J]. 广东农业科学, 2012, 39(8): 124−127. doi: 10.3969/j.issn.1004-874X.2012.08.039Shu Hu, Wei Qiulan, Luo Lijuan, et al. Karyotypes analysis of four grouper fishes from coastal waters of Guangdong[J]. Guangdong Agricultural Sciences, 2012, 39(8): 124−127. doi: 10.3969/j.issn.1004-874X.2012.08.039 [18] 刘苏, 杨宇晴, 张海发, 等. 蓝身大斑石斑鱼染色体核型分析[J]. 海洋科学, 2017, 41(12): 46−50. doi: 10.11759/hykx20170613002Liu Su, Yang Yuqing, Zhang Haifa, et al. The karyotype of Epinephelus tukula[J]. Marine Sciences, 2017, 41(12): 46−50. doi: 10.11759/hykx20170613002 [19] 李振通, 成美玲, 田永胜, 等. 杂交种“云龙石斑鱼”与亲本的形态差异分析[J]. 渔业科学进展, 2019, 40(4):73−83.Li Zhentong, Cheng Meiling, Tian Yongsheng, et al. Analysis of the morphological differences between hybrid Epinephelus moara ♀× E. lanceolatus♂ and its parents[J]. Progress in Fishery Sciences, 2019, 40(4):73−83. [20] 唐江, 田永胜, 李振通, 等. 云纹石斑鱼和鞍带石斑鱼及其杂交后代遗传性状分析[J]. 农业生物技术学报, 2018, 26(5): 819−829.Tang Jiang, Tian Yongsheng, Li Zhentong, et al. Analysis of genetic characters in Epinephelus moara, E. lanceolaus and their hybrids[J]. Journal of Agricultural Biotechnology, 2018, 26(5): 819−829. [21] 刘筠. 中国养殖鱼类繁殖生理学[M]. 北京: 农业出版社, 1993: 109-124.Liu Jun. Propagation Physiology of Main Cultivated Fish in China[M]. Beijing: Agriculture Press, 1993: 109-124. [22] Hodson P V, Blunt B R. The effect of time from hatch on the yolk conversion efficiency of rainbow trout, Salmo gairdneri[J]. Journal of Fish Biology, 1986, 29(1): 37−46. doi: 10.1111/jfb.1986.29.issue-1 [23] Avila E M, Juario J V. Yolk and oil globule utilization and developmental morphology of the digestive tract epithelium in larval rabbitfish, Siganus guttatus (Bloch)[J]. Aquaculture, 1987, 65(3/4): 319−331. [24] Kohno H, Hara S, Taki Y. Early larval development of the seabass Lates calcarifer with emphasis on the transition of energy sources[J]. Bulletin of the Japanese Society of Scientific Fisheries, 1986, 52: 1719−1725. doi: 10.2331/suisan.52.1719 [25] Nash C E, Kuo C M, McConnel S C. Operational procedures for rearing larvae of the grey mullet (Mugil cephalus L.)[J]. Aquaculture, 1974, 3(1): 15−24. doi: 10.1016/0044-8486(74)90095-7 [26] 谢菁, 区又君, 李加儿, 等. 七带石斑鱼胚体和卵黄囊期仔鱼的发育[J]. 海洋通报, 2009, 28(2): 41−49. doi: 10.3969/j.issn.1001-6392.2009.02.007Xie Jing, Ou Youjun, Li Jiaer, et al. Development of neural embryo and yolk sac larvae of Epinephelus septemfasciatus[J]. Marine Science Bulletin, 2009, 28(2): 41−49. doi: 10.3969/j.issn.1001-6392.2009.02.007 [27] 张俊玲, 施志仪. 牙鲆早期阶段的变态发育及其机制[J]. 上海水产大学学报, 2003, 12(4): 348−352.Zhang Junling, Shi Zhiyi. Metamorphosis and mechanism during early development of Japanese flounder, Paralichthys olivaceus[J]. Journal of Shanghai Fisheries University, 2003, 12(4): 348−352. [28] Shao Changwei, Bao Baolong, Xie Zhiyuan, et al. The genome and transcriptome of Japanese flounder provide insights into flatfish asymmetry[J]. Nature Genetics, 2017, 49(1): 119−124. doi: 10.1038/ng.3732 [29] 唐啸尘, 刘晓春, 林浩然. 斜带石斑鱼仔鱼变态过程中甲状腺的发育变化[J]. 水生生物学报, 2010, 34(1): 210−214.Tang Xiaochen, Liu Xiaochun, Lin Haoran. The development of thyroid of orange-spotted grouper (Epinephelus coioides) larvae during metamorphosis[J]. Acta Hydrobiologica Sinica, 2010, 34(1): 210−214. -




 下载:
下载: